74. M-N-C Cluster Decorated TiC is a Durable and Inexpensive ORR Electrocatalyst
Sung Beom Cho et al. ChemSusChem (2021)
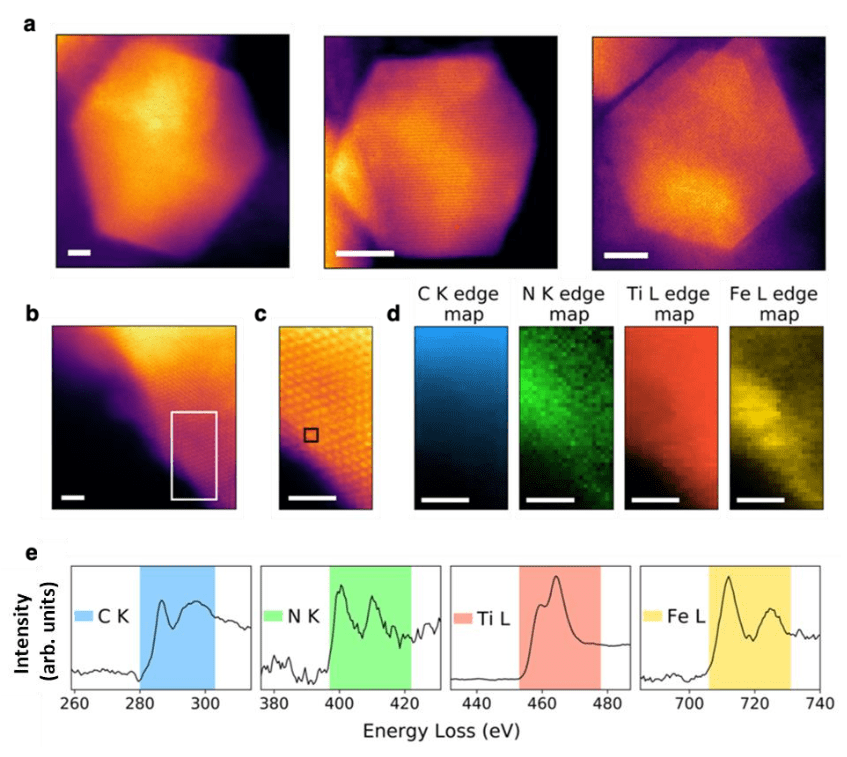
Metal-Nitrogen-Carbon Cluster Decorated Titanium Carbide is a Durable and Inexpensive Oxygen Reduction Reaction Electrocatalyst
Sung Beom Cho, Cheng He, Shrihari in Sankarasubramanian, Arashdeep Singh Thind, Javier Parrondo, Jordan A. Hachtel, Albina Y. Borisevich, Juan-Carlos Idrobo, Jing Xie, Vijay K. Ramani, Rohan Mishra, ChemSusChem(2021)
Clusters of nitrogen- and carbon-coordinated transition metals dispersed in a carbon matrix (e.g. Fe-N-C) have emerged as an inexpensive class of electrocatalysts for the oxygen reduction reaction (ORR). Here we show that optimizing the interaction between the nitrogen-coordinated transition metal clusters embedded in a more stable and corrosion-resistant carbide matrix yields an ORR electrocatalyst with enhanced activity and stability compared to Fe-N-C catalysts. Utilizing first-principles calculations, we identify an electrostatics-based descriptor of catalytic activity and predict nitrogen-coordinated iron (FeN4) clusters embedded in a TiC matrix to be an efficient PGM-free ORR electrocatalyst. Guided by theory, we synthesized selected catalyst formulations and demonstrated that the experimentally observed trends in activity fall exactly in line with our descriptor-derived theoretical predictions. The Fe-N-TiC catalyst exhibited enhanced activity (20%) and durability (3.5-fold improvement) compared to a traditional Fe-N-C catalyst. We posit that the electrostatics-based descriptor provides a powerful platform for the design of active and stable PGM-free electrocatalysts and heterogenous single-atom catalysts for other electrochemical reactions.