18. Origin of barriers for ionic transport through interfaces
M. Frechero et al., Scientific Reports 5, 17229 (2015).
Paving the way to nanoionics: atomic origin of barriers for ionic transport through interfaces.
M. Frechero, M. Rocci, G. Sanchez-Santolino, A. Kumar, J. Salafranca, R. Schmidt, M. Diaz-Gullen, O. Dura, A. Rivero-Calzada, R. Mishra, S. Jesse, S. T. Pantelides, S. V. Kalinin, M. Varela, S. J. Pennycook, J. Santamaria, C. Leon, Scientific Reports 5, 17229 (2015).
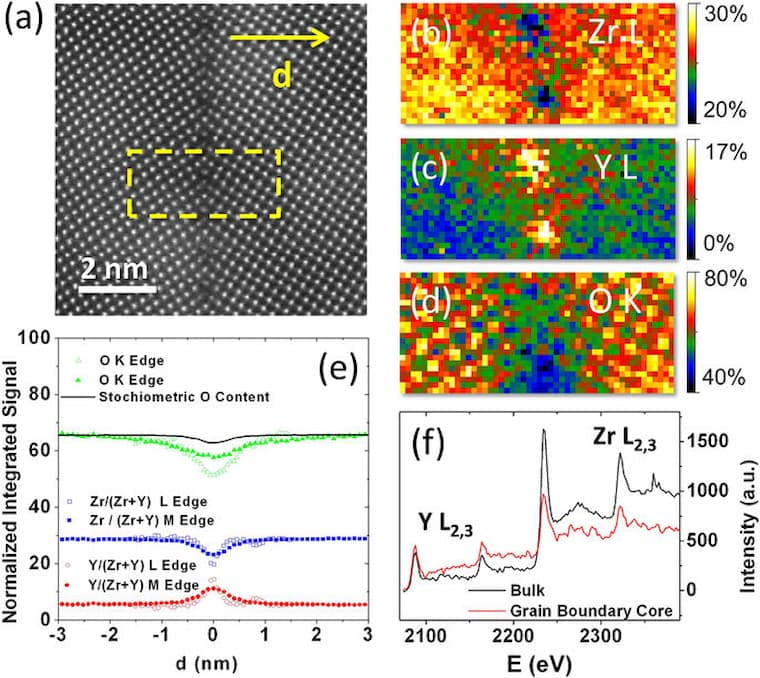
The blocking of ion transport at interfaces strongly limits the performance of electrochemical nanodevices for energy applications. The barrier is believed to arise from space-charge regions generated by mobile ions by analogy to semiconductor junctions. Here we show that something different is at play by studying ion transport in a bicrystal of yttria (9% mol) stabilized zirconia (YSZ), an emblematic oxide ion conductor. Aberration-corrected scanning transmission electron microscopy (STEM) provides structure and composition at atomic resolution, with the sensitivity to directly reveal the oxygen ion profile. We find that Y segregates to the grain boundary at Zr sites, together with a depletion of oxygen that is confined to a small length scale of around 0.5 nm. Contrary to the main thesis of the space-charge model, there exists no evidence of a long-range O vacancy depletion layer. Combining ion transport measurements across a single grain boundary by nanoscale electrochemical strain microscopy (ESM), broadband dielectric spectroscopy measurements, and density functional calculations, we show that grain-boundary-induced electronic states act as acceptors, resulting in a negatively charged core. Besides the possible effect of the modified chemical bonding, this negative charge gives rise to an additional barrier for ion transport at the grain boundary.